- Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
-
Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
MR/US fusion aligns and registers prior diagnostic MR images with real-time ultrasound images. UroNav displays the segmented prostate anatomy from the MR exam, indicated by the purple outline, while green and red “targets” indicate the location of the MR suspicious lesions. - View clinical data prior to biopsy for added confidence
-
View clinical data prior to biopsy for added confidence
With the ability to preview MR images, gland segmentation, biopsy “targets”, and PI-RADS® information from Radiology prior to the biopsy, Urologists can perform biopsy procedures with added confidence. - Support for both transrectal and transperineal biopsy
-
Support for both transrectal and transperineal biopsy
UroNav supports both transrectal and transperineal (stepper or freehand) biopsy approaches, providing users the flexibility necessary to incorporate fusion-guided biopsy into their preferred biopsy method. - Comprehensive, 3D review of the biopsy following the procedure
-
Comprehensive, 3D review of the biopsy following the procedure
The clinical team can review biopsy targets and core samples taken during the procedure in 3D, which assists users in easily identifying biopsy sample locations. UroNav also provides users with access to view and export screen captures and video recording taken during the fusion-guided biopsy. - UroNav can guide your biopsy – even without pre-biopsy MR images
-
UroNav can guide your biopsy – even without pre-biopsy MR images
Ultrasound-only workflow with UroNav allows for tracking of biopsy locations under ultrasound guidance without a prior MRI exam. Biopsy locations can be registered with a future MRI and then targeted for resampling. Ultrasound-only procedure data, including biopsy location data, can be exported to DynaCAD Prostate for further review and analysis. - Current/Prior fusion displays points of reference during biopsies
-
Current/Prior fusion displays points of reference during biopsies
UroNav automatically displays biopsy locations from prior procedures. These locations can be used as points of reference during the MR fusion biopsy procedure. Users can review and adjust the prior-to-current registration to include an optional image-based elastic deformation.
Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
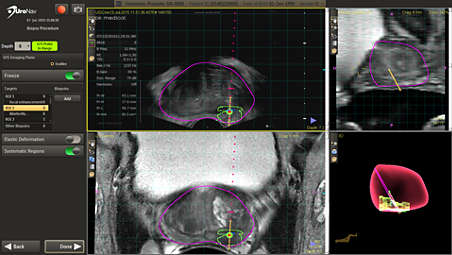
Fuse pre-biopsy MR and ultrasound images for a targeted biopsy

Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
View clinical data prior to biopsy for added confidence
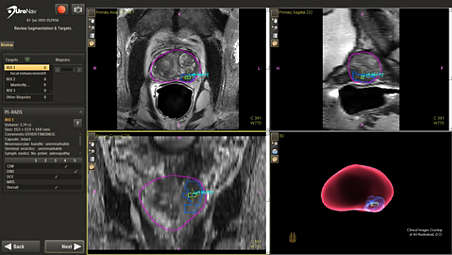
View clinical data prior to biopsy for added confidence

View clinical data prior to biopsy for added confidence
Support for both transrectal and transperineal biopsy
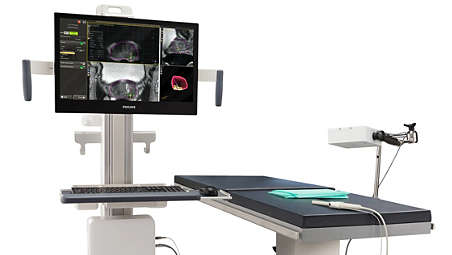
Support for both transrectal and transperineal biopsy

Support for both transrectal and transperineal biopsy
Comprehensive, 3D review of the biopsy following the procedure
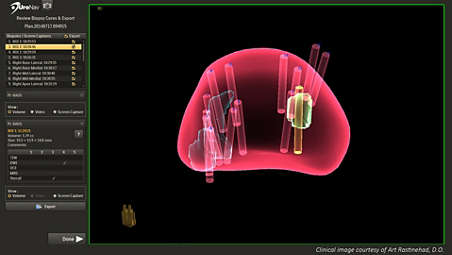
Comprehensive, 3D review of the biopsy following the procedure

Comprehensive, 3D review of the biopsy following the procedure
UroNav can guide your biopsy – even without pre-biopsy MR images
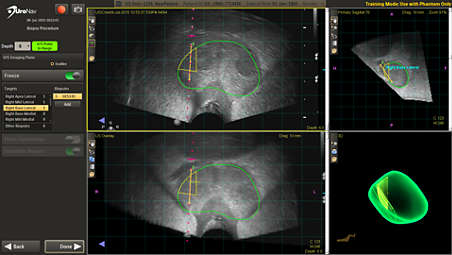
UroNav can guide your biopsy – even without pre-biopsy MR images

UroNav can guide your biopsy – even without pre-biopsy MR images
Current/Prior fusion displays points of reference during biopsies
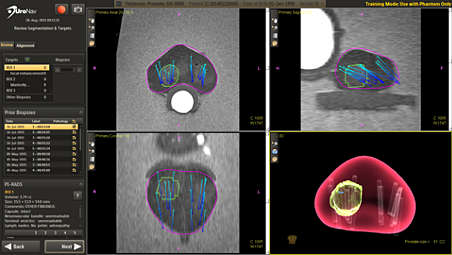
Current/Prior fusion displays points of reference during biopsies

Current/Prior fusion displays points of reference during biopsies
- Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
- View clinical data prior to biopsy for added confidence
- Support for both transrectal and transperineal biopsy
- Comprehensive, 3D review of the biopsy following the procedure
- Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
-
Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
MR/US fusion aligns and registers prior diagnostic MR images with real-time ultrasound images. UroNav displays the segmented prostate anatomy from the MR exam, indicated by the purple outline, while green and red “targets” indicate the location of the MR suspicious lesions. - View clinical data prior to biopsy for added confidence
-
View clinical data prior to biopsy for added confidence
With the ability to preview MR images, gland segmentation, biopsy “targets”, and PI-RADS® information from Radiology prior to the biopsy, Urologists can perform biopsy procedures with added confidence. - Support for both transrectal and transperineal biopsy
-
Support for both transrectal and transperineal biopsy
UroNav supports both transrectal and transperineal (stepper or freehand) biopsy approaches, providing users the flexibility necessary to incorporate fusion-guided biopsy into their preferred biopsy method. - Comprehensive, 3D review of the biopsy following the procedure
-
Comprehensive, 3D review of the biopsy following the procedure
The clinical team can review biopsy targets and core samples taken during the procedure in 3D, which assists users in easily identifying biopsy sample locations. UroNav also provides users with access to view and export screen captures and video recording taken during the fusion-guided biopsy. - UroNav can guide your biopsy – even without pre-biopsy MR images
-
UroNav can guide your biopsy – even without pre-biopsy MR images
Ultrasound-only workflow with UroNav allows for tracking of biopsy locations under ultrasound guidance without a prior MRI exam. Biopsy locations can be registered with a future MRI and then targeted for resampling. Ultrasound-only procedure data, including biopsy location data, can be exported to DynaCAD Prostate for further review and analysis. - Current/Prior fusion displays points of reference during biopsies
-
Current/Prior fusion displays points of reference during biopsies
UroNav automatically displays biopsy locations from prior procedures. These locations can be used as points of reference during the MR fusion biopsy procedure. Users can review and adjust the prior-to-current registration to include an optional image-based elastic deformation.
Fuse pre-biopsy MR and ultrasound images for a targeted biopsy

Fuse pre-biopsy MR and ultrasound images for a targeted biopsy

Fuse pre-biopsy MR and ultrasound images for a targeted biopsy
View clinical data prior to biopsy for added confidence

View clinical data prior to biopsy for added confidence

View clinical data prior to biopsy for added confidence
Support for both transrectal and transperineal biopsy

Support for both transrectal and transperineal biopsy

Support for both transrectal and transperineal biopsy
Comprehensive, 3D review of the biopsy following the procedure

Comprehensive, 3D review of the biopsy following the procedure

Comprehensive, 3D review of the biopsy following the procedure
UroNav can guide your biopsy – even without pre-biopsy MR images

UroNav can guide your biopsy – even without pre-biopsy MR images

UroNav can guide your biopsy – even without pre-biopsy MR images
Current/Prior fusion displays points of reference during biopsies

Current/Prior fusion displays points of reference during biopsies

Current/Prior fusion displays points of reference during biopsies
Documentation
-
Brochure (1)
-
Brochure
- Product brochure (1.8 MB)
-
Brochure (1)
-
Brochure
- Product brochure (1.8 MB)
-
Brochure (1)
-
Brochure
- Product brochure (1.8 MB)
Specifications
- UroNav Cart Dimensions
-
UroNav Cart Dimensions Weight - 69.7 kg
Display - 24 in diagonally
System Dimensions - 20.5D x 31.5W x 66.5H inches
-
- UroNav Cart Dimensions
-
UroNav Cart Dimensions Weight - 69.7 kg
Display - 24 in diagonally
-
- UroNav Cart Dimensions
-
UroNav Cart Dimensions Weight - 69.7 kg
Display - 24 in diagonally
System Dimensions - 20.5D x 31.5W x 66.5H inches
-
Related products
Alternative products
-
DynaCAD Prostate
- Post-processing image analysis
- Standardized reporting
- Access virtually anywhere
View product
-
DynaCAD Urology
- View MR images and segmentation data
- Plan treatment steps and follow-up
- Gain time and workflow efficiencies
- Enhance confidence for biopsies
View product
-
DynaCAD Prostate
Philips DynaCAD Prostate is an advanced visualization system that empowers you with a comprehensive set of tools for real-time analysis, review, and reporting of multi-parametric MRI studies. Create time and workflow efficiency by transferring images directly from the MRI to DynaCAD. Utilize its robust, automatic post-processing tools and display results in customized hanging protocols for analysis and reporting. At case completion, you can automatically transfer key images, statistical data, and prostate PI-RADS® reports to PACS for archiving. By setting everything up for you to work, DynaCAD helps you enhance your confidence and productivity – so patients get the prompt, precise care they need.
View product
-
DynaCAD Urology
DynaCAD Urology is a purpose build solution that empowers urologists with a dedicated set of tools for utilizing multi-parametric MR data in fusion biopsy workflows. It also provides a solution for managing patients’ biopsy data in urology.
View product
- PI-RADS® is a registered trademark of The American College of Radiology.
- Product not available for sale in all countries. Please contact your sales representative to ascertain availability in your country.